Some interesting and informative demonstrations are prepared as a useful teaching tool to explain the idea of energy quantization. One of the demonstrations is made using a tube filled with low-pressure hydrogen and observing therefrom the line spectrum. Another demonstration presents the continuous spectrum from an incandescent tungsten filamented lamp as a source of white light. One can use it to distinguish between continuous and line spectrums. One can also optically filter light of desired wavelengths. The spectrums from red, green, blue and white LEDs are also shown. Last, a mercury filled tube has a more complicated line spectrum which students find appealing.
Spectrum of Different Elements using Spectrometer
- Setup for observing the line spectrum of Mercury
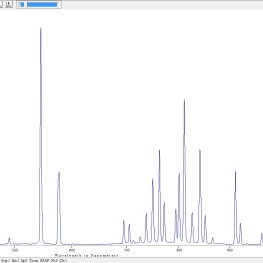
- Line spectrum of Mercury
- Setup for observing the line spectrum of Hydrogen
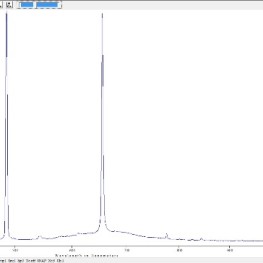
- Hydrogen spectrum
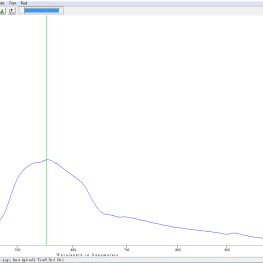
- Spectrum of incandescent tungsten bulb
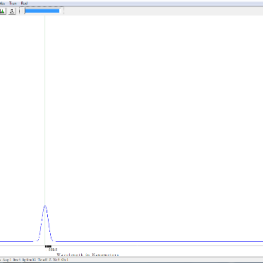
- incandescent tungsten bulb spectrum with green filter